PRESS and NEWS
-------------------------------
- 2026.2.4 | Clinical Pharmacology | DMPK
Save the date: March 26, 2026; Peregrion B.V. organizes its 2nd AMS symposium.
Today, on World Cancer Day, we're reminded why speed matters in drug development. Nearly 45% of new drugs in development target oncology and immunology. For every cancer patient waiting, time is measured not in quarters but in quality of life.
The traditional path? Wait until Phase 2b/3 to understand how a drug behaves in the human body. By then, you've invested years and millions, only to discover metabolic surprises that could have been caught much earlier.
At Peregrion, we're changing this timeline. Our microtracer AMS technology enables pharmaceutical teams to obtain complete human ADME data in Phase 1.
This means:
→ Identifying metabolite safety concerns when adjustments are still possible
→ Making go/no-go decisions years earlier
→ Getting promising therapies to patients up to 2 years faster
For cancer patients, two years isn't just a statistic. It's time with family. It's treatment options. It's hope. This World Cancer Day, we're committed to accelerating the path from lab to life-saving therapy.
Location: Fletcher Wellness-Hotel Leiden
Address:Bargelaan 180, 2333 CW Leiden
Registration: Sign up now

- 2026.1.29 | Translational Research | Metabolic Health
Two TNO researchers, Anita M. van den Hoek and Reinout Stoop, presented the following poster at the Keystone Symposia, Cardiometabolism in Health and Disease, held in Keystone, Colorado, on January 28th.
□ Treatment with either SGLT2-inhibition or GLP1Ra Treatment Rescues GFR Decline in a Multifactorial Cardiovascular-Kidney-Metabolic Syndrome Mouse Model.
□ Semaglutide and Exercise Synergy in Obesity: Preserving Muscle Mass and Uncovering Organ Crosstalk.

- 2025.11.17 | Translational Research | Metabolic Health
In early November, TNO presented at several major international conferences focusing on obesity, kidney, heart and liver diseases, provided an excellent opportunity for the pharmaceutical and biotechnology industries to discover how TNO's unique strengths in preclinical translational research can support their drug discovery and innovation goals.
Obesity Week in Atlanta, November 4-7 (2 posters)
□ Obesity accelerates age-related memory deficits and affects brain connectivity and white matter tract integrity in Ldlr-/-.Leiden mice.
□ Exercise Partly Protects Against Semaglutide-Induced Muscle Loss in Obese Ldlr-/-.Leiden Mice.
ASN Kidney Week in Houston, November 6-9 (1 oral presentation)
□ Treatment with Either SGLT2 Inhibitors or GLP-1 Receptor Agonists Rescues GFR Decline in a Multifactorial Cardiovascular-Kidney-Metabolic Syndrome Mouse Model.
Scientific Sessions of the American Heart Association in New Orleans, November 7-10 (1 poster)
□ Obicetrapib in combination with ezetimibe on top of atorvastatin regresses atherosclerotic plaque lesions in APOE*3-Leiden.CETP mice.
The Liver Meeting in Washington, November 7-11 (3 posters)
□ Female sex hormones are negatively associated with metabolic dysfunction-associated steatotic liver disease in children with overweight and obesity.
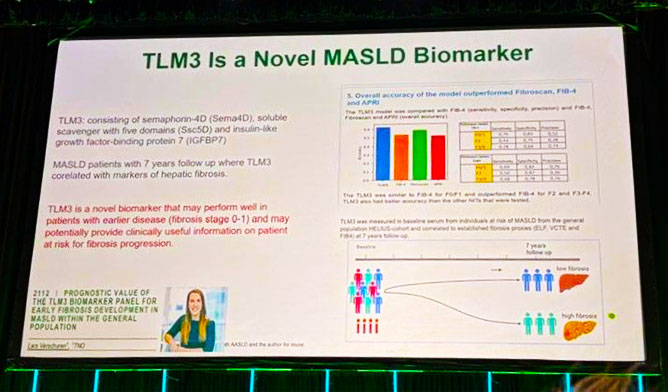
□ Prognostic value of the TLM3 biomarker panel for early fibrosis development in MASLD within the general population.
□ Exercise Partly Protects Against Semaglutide-Induced Muscle Loss in Obese Ldlr-/-.Leiden Mice.
★ Our poster on the TLM3 biomarker for MASLD fibrosis highlighted in the TLM Debrief on Basic Science, Hepatitis, HCC, Cholestatic, MASLD, Alcohol-Associated Liver Disease, and MetALD at AASLD’s Liver Meeting in Washington.
- 2025.10.24 | Clinical Pharmacology | DMPK
TNO presented the following two posters at the 40th Annual Meeting of the Japanese Society for the Study of Xenobiotics held at Miyako Messe in Kyoto from October 20th to 23rd.
□ Normothermic machine perfusion of ex vivo porcine liver: Demonstrator studies for preclinical testing of an RNA therapeutic and multi-day perfusion feasibility. (P-127)
□ Advancing Drug Research with the InTESTineTM Platform: Ex Vivo Intestinal explant Applications for Peptides and Permeability Enhancers. (P-076)

- 2025.10.1 | Clinical Pharmacology | DMPK
A paper has been published in the Antimicrobial Agents and Chemotherapy reporting on a human mass balance study of Acoziborole, a promising single dose oral treatment of human African trypanosomiasis, a deadly disease. TNO, Peregrion (Leiden, the Netherlands) performed the analysis of total radioactivity and the metabolite profiling and identification.
□ Mass balance, pharmacokinetics, metabolism, and excretion of radiolabeled acoziborole, a potential novel treatment for human African trypanosomiasis, following single microtracer oral dose to humans
- 2025.8.20 | Metabolic Health
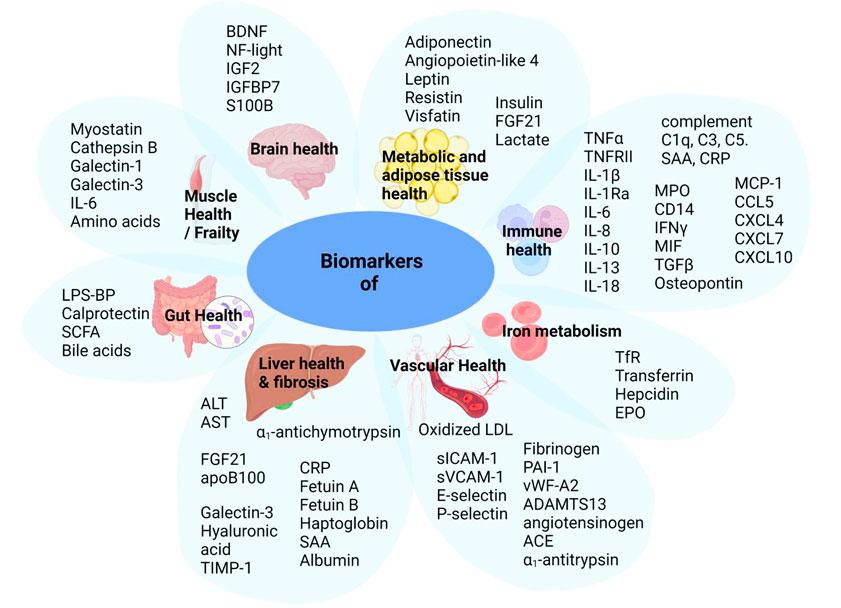
TNO has published a research paper in the International Journal of Molecular Science entitled Biomarkers of Metabolism and Inflammation in Individuals with Obesity and Normal Weight: A Comparative Analysis Exploring Sex Differences. In this study, we analyzed over 90 biomarkers from healthy normal-weight and obese individuals using ELISA and electrochemiluminescence. The results revealed that many biomarkers associated with chronic inflammation and metabolic disorders, such as adipokines and cytokines, were elevated in obese individuals. The study also suggested potential sex differences in biomarker levels in both groups. This dataset not only assists researchers in selecting appropriate biomarkers for their own studies but also highlights the importance of considering sex differences in future research.
- 2025.7.29 | DMPK
TNO's research on an ex vivo new multi-organ perfusion model that reproduces conditions similar to those in vivo by circulating blood and perfusion fluid through the vascular systems of organs such as the liver, intestines and kidneys, won the Best Paper of the Year Award 2024 from the European Journal of Pharmaceutical Sciences. Link
□ Ex vivo gut-hepato-biliary organ perfusion model to characterize oral absorption, gut-wall metabolism, pre-systemic hepatic metabolism and biliary excretion; application to midazolam
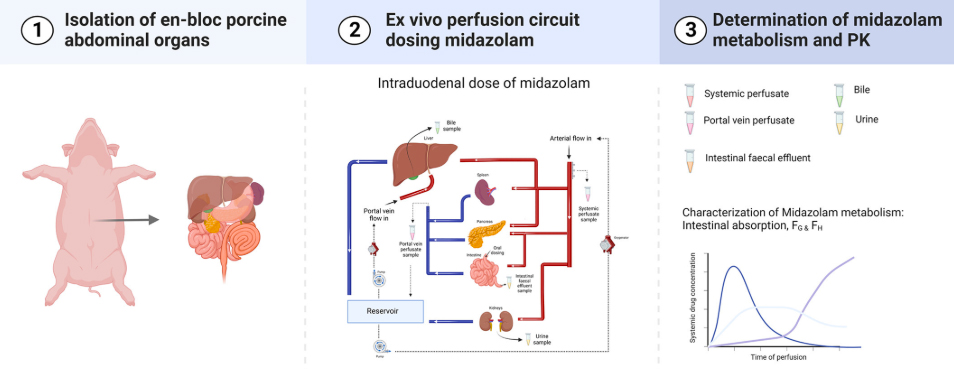
- 2025.5.10 | Metabolic Health
TNO presented the following two research results in poster at EASL congress 2025 held in Amsterdam from May 7th to 10th.
□ Exercise partly protects against semaglutide-induced muscle loss in obese Ldlr-/-.Leiden mice.
□ Anti-inflammatory effects of a chemokine receptor mimicking peptide in obesity-associated MASH and atherosclerosis in Ldlr-/- Leiden mice.

- 2025.3.7| Metabolic Health
TNO scientists participate the 15th International Conference on Frailty and Sarcopenia Research at Toulouse, France from March 12th to 14th, and present the following two research results
□ Exercise protects against semaglutide-induced muscle loss in obese Ldlr-/-.Leiden mice. (March 13, 11:20 AM)
□ Meta-analysis reveals muscle-aging processes are generally shared between females and males at the transcriptome level (Poster)

- 2025.3.6 | Metabolic Health
For over 20 years, TNO’s ApoE*3-Leiden.CETP mouse model has proven to be the most translational model in atherosclerosis research, as evidenced by numerous publications in esteemed, peer-reviewed journals. The journey continues! Recently, Jose Inia et al. published a groundbreaking paper titled “Efficacy of a novel PCSK9 inhibitory peptide alone and with evinacumab in a mouse model of atherosclerosis” in the Journal of Lipid Research. In a previous study, we demonstrated the translational value of the ApoE*3-Leiden.CETP model for evinacumab, highlighting its role in elucidating the underlying processes and mechanisms. The current study’s results suggest that Novo Nordisk’s PCSK9 inhibitory peptide offers a promising approach for patients with atherosclerotic CVD who struggle to meet LDL-C targets with conventional statins or are statin-intolerant. Read the full paper here:

- 2025.2.14 | Metabolic Health
TNO scientists will be participating in Obesity and Adipose Tissue (MASH Pathogenesis and Therapeutic Approaches) to be held at Fairmont Banff Springs, Banff, AB, Canada from February 23rd to 26th, and will present the following two posters.
□ 25 Feb: Resmetirom protects against diet-induced MASLD and reduces atherosclerosis development in obese Ldlr-/-.Leiden mice. (#2519)
□ 26 Feb: TLM3 biomarker panel is prognostic for early MASLD fibrosis development in the general population. (#3052)
- 2025.2.13 | Microbiology, Oral Health
A paper by our Microbiology scientists "Yeast Cell Wall Derivatives as a Potential Strategy for Modulating Oral Microbiota and Dental Plaque Biofilm," has been provisionally accepted by Frontiers. Key Findings: Yeast cell wall derivatives significantly reduced biofilm density and promoted a beneficial shift in microbial composition. Decreased abundance of Tannerella forsythia, a key species in biofilm development. Lowered production of short-chain fatty acids like acetic and butyric acid, positively impacting both healthy and periodontitis microbiomes. These results underline the potential of yeast-based compounds as a sustainable and innovative solution for managing oral health.
□ Yeast cell wall derivatives as a potential strategy for modulating oral microbiota and dental plaque biofilm.
- 2024.11.28 | Metabolic Health
TNO scientists presented the latest findings in MASH research as the followings at AASLD The 75th Liver Meeting in San Diego.
□ Resmetirom protects against diet-induced MASLD and reduces atherosclerosis development in obese Ldlr-/-.Leiden mice.
□ Metabolic flux analysis using a 14C microtracer approach combined with accelerator mass spectrometry analysis - a proof of concept study on de novo lipogenesis.
□ Treatment with the CCR2/CCR5 antagonist Cenicriviroc does not affect MASH and fibrosis development in Ldlr-/-.Leiden mice, translational to clinical phase 3 trial results.
□ Novel blood-based biomarker model for diagnosis of hepatic fibrosis in MASLD.

- 2024.10.30 | General
TNO and the Japanese AIST Group (National Institute of Advanced Industrial Science and Technology (AIST) and AIST Solutions Co (AISol) signed a Memorandum of Understanding (MOU) to expand their existing cooperation across a wide range of technological fields. Related post Link;TNO AIST

- 2024.10.29 | Translational Research
A new paper on the MASH model Ldlr-/-.Leiden mice is published in the FASEB Journal. FGF21 is a promising target for the treatment of obesity-related diseases such as MASH and atherosclerosis, and the pharmacological effect of bispecific anti-FGF21-β klotho (KLB) agonist antibody bFKB1 was evaluated in this model.
□ Therapeutic effects of FGF21 mimetic bFKB1 on MASH and atherosclerosis in Ldlr-/-.Leiden mice
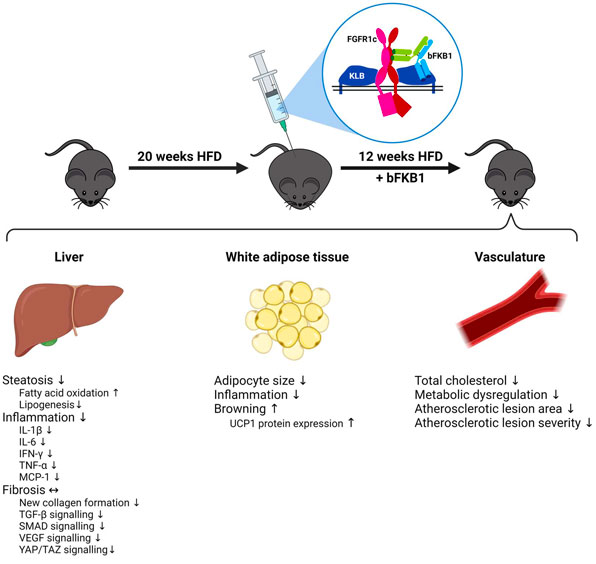
- 2024.7.12 | Translational Research
Another groundbreaking paper on TNO’s biomarkers has been published in a relevant scientific journal: Hogenelst et al. unveiled seven robust and easy-to-obtain biomarkers to measure acute stress in Brain Behavior and Immunity - Health, July 2024. This research focuses on functional biomarkers for stress, surpassing traditional cortisol measurements. It enhances our understanding of stress effects on metabolic health and offers new avenues for stress quantification. These findings are pivotal for stress management interventions, fostering a healthier society. Publication link
- 2024.7.10 | Clinical Pharmacology | DMPK
Peregrion B.V., a 100% subsidiary of TNO specializing in human ADME studies using accelerator mass spectrometry (AMS) and 14C microtracers, has started its contract analysis service as of July 1st. Peregrion B.V. will continue TNO's track record of generating early human data from human ADME studies. TNO has a long history of process optimization, for example developing a fully automated analytical system that can detect, identify and quantify all human metabolites at the early stage of clinical phase I. Currently, the top 20 major pharmaceutical companies are promoting drug development programs using this AMS technology, and industry acceptance is expanding. Press Release link
- 2024.5.29 | Translational Research
Development of a novel non-invasive biomarker panel for hepatic fibrosis in MASLD, which is being promoted by TNO, has been published in Nature Communication
□ Development of a novel non-invasive biomarker panel for hepatic fibrosis in MASLD
- 2024.4.8 | Translational Research
New webpage for in vivo MASH (NASH) model, Liver on a Chip (Cell model for Liver fibrosis) and MASH Biomarkers is introduced in this website. Link
TNO has developed the Ldlr-/-.Leiden MASH mouse model that accurately mimics the etiology and pathology of MASH and fibrosis in humans. By using a high-fat diet, with a macronutrient composition comparable to human diets (e.g., without added cholesterol), the Ldlr-/-.Leiden MASH mouse develops obesity, insulin resistance, adipose tissue inflammation, increased gut permeability with altered microbiota composition, and MASH with bridging fibrosis [F3 stage]. The Ldlr-/-.Leiden MASH mouse has extensive validation records with multiple treatments and drugs, including Semaglutide (GLP-1) and Rezdiffra (Resmetirom) approved by FDA in March 2024.
- 2024.3.22 | Microbiology, DMPK
A recent study published in Microbiome Research Report 2024 has demonstrated the beneficial effects of inulin on the gut microbiota using a TNO Gut-on-a-Chip model of the adult human intestine that recapitulates host-microbe metabolite interactions.
The results showed that inulin administration improved the balance of gut bacteria and had a positive impact on health. Publication Link: A host-microbial metabolite interaction gut-on-achip model of the adult human intestine demonstrates beneficial effects upon inulin treatment of gut microbiome
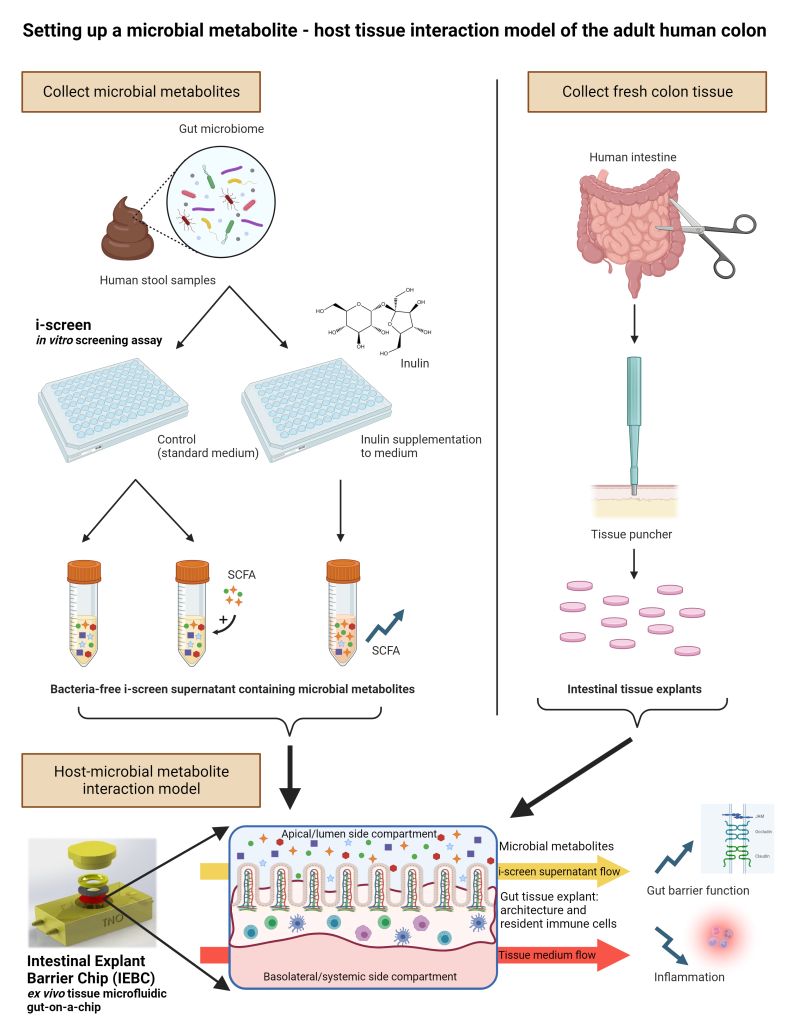
- 2024.3.3 | Metabolic Health
TNO scientists, Reinout Stoop and Arianne van Koppen participate the 6th CKD Summit in Boston on 20 and 21 March.
They will present their poster on TNO's Cardiovascular Kidney Metabolic Syndrome mouse model with CKD and HFpEF.

- 2024.1.12 | Digital HealthTNO joins the iCARE4CVD consortium for personalised cardiovascular care; TNO is one of 33 international partners that have joined forces in the research consortium iCARE4CVD, to better understand cardiovascular disease and optimise future prevention and treatment. By creating one database consisting of data from more than 1 million patients and using artificial intelligence, partners will look for new strategies to shift from a one-size-fits-all approach to personalised care. The consortium brings together international partners from society, academia, and industry, led and coordinated by Maastricht University and Novo Nordisk. Link to Digital Biomarker